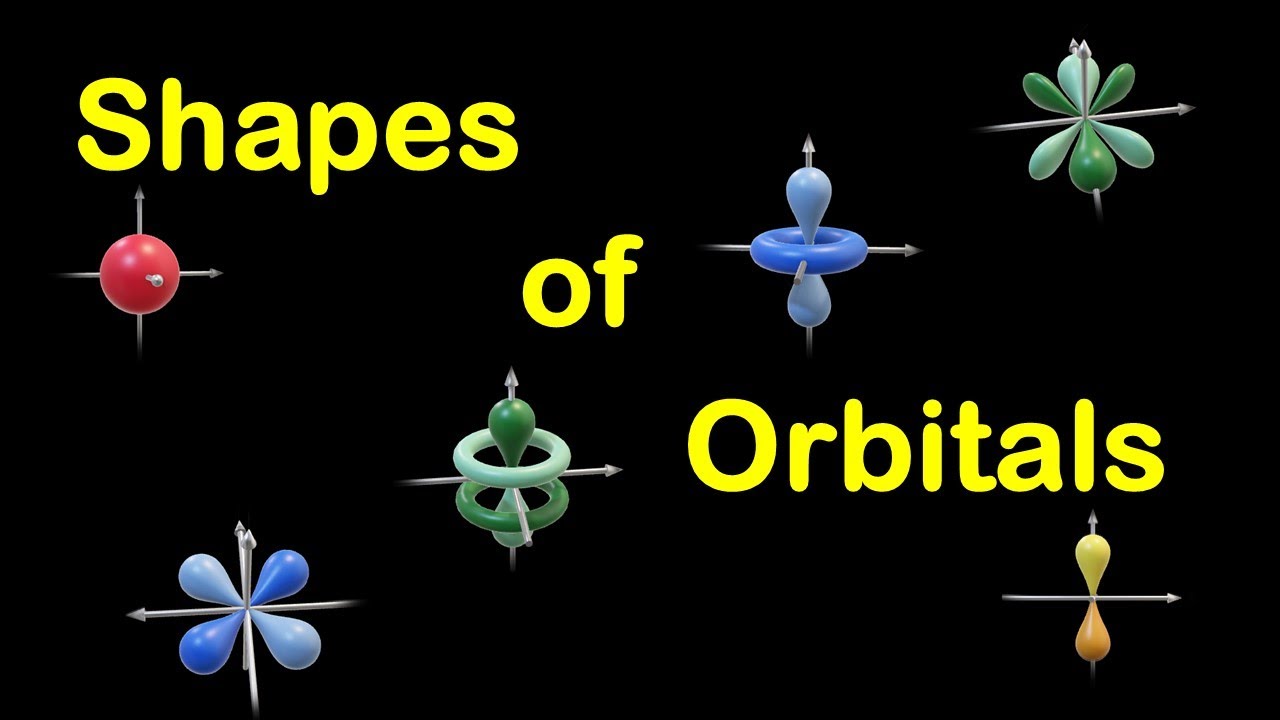
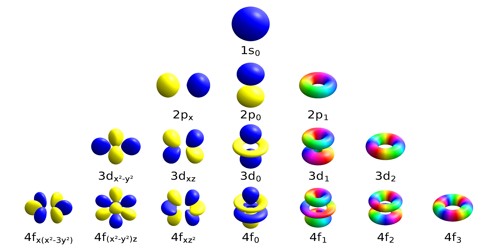
  Like an atomic orbital, a molecular orbital can hold a maximum number of two electrons. According to Aufbau rule, the molecular orbitals are filled from low energy orbital to high energy orbital. The final shape of the molecular orbital depends on the shapes of atomic orbitals that make the molecule. Thus, the nuclei of different atoms behave as a polycentric nucleus. Unlike atomic orbitals, the molecular orbitals do not belong to a single atom in a molecule but belong to nuclei of all the atoms which make the molecule. Molecular orbitals are formed by the combination of nearly the same energy atomic orbitals. The new orbitals present in the molecules are now called molecular orbitals. According to molecular orbital theory, when atoms are fused to form a molecule, the overlapping atomic orbitals lose their shape due to the effect of nuclei. The properties of molecular orbitals are explained by the molecular orbital theory. The energies of orbitals are in order of s  The s orbital is spherical and hold a maximum of two electrons. Atomic orbitals are labeled as s, p, d, and f sublevels. An atomic orbital can have a maximum of two electrons. The electron density of an atom can be found from the solutions of the Schrodinger equation. It is explained in Heisenberg’s uncertainty principle. It does not explain the exact energy of an electron at a given prompt of time. Quantum mechanics explain the probability of the location of an electron of an atom. The Atomic orbital is a region having the highest probability of finding an electron. What is the difference between Atomic Orbital and Molecular Orbital – Definition, Characteristics, Propertiesģ. The main difference between atomic and molecular orbital is that the electrons in an atomic orbital are influenced by one positive nucleus, while the electrons of a molecular orbital are influenced by the two or more nuclei depending upon the number of atoms in a molecule. Orbitals can hold a maximum of two electrons. Valence bond theory and molecular orbital theory explains the properties of atomic and molecular orbitals, respectively. When these orbitals are overlapped to form molecules through the bonding, the orbitals are called molecular orbitals. Atoms have their own electrons rotating around the nucleus. Orbital is defined as a region where the probability of finding an electron is high. This is also the case with methane, where px, py-s overlapping occurs, assuming that in sp3-hybridized state px, py orbitals in C bond with H 1s orbitals.Main Difference – Atomic Orbital vs Molecular Orbital Thus s and px and s and py orbitals do overlap if you take px to be of lower energy than py and pz, s-px overlapping occurs in boranes (formed by boron, which has one electron in 2px orbital, and hydrogen, which has one electron in 1s orbital), that often dimerize. As s orbitals are spherical, they have infinite possibilities of orbital overlap with other orbital shapes and thus even overlap with d orbitals (as explained by VSEPR: Valence Shell Electron Pair Repulsion Theory in a similar manner). Your instructor might be pointing out to the bond-symmetry thing in MO formation between pz and s orbitals extrapolated in most chemistry textbooks.īut orbital overlap is a result of constructive and destructive interference. "Molecular orbitals obtained from 2px and 2py orbitals are not symmetrical around the bond axis because of the positive lobes above and below the molecular plane."
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
